Effects of Freezing Conditions on Colloidal Silica
What happens when the binder is kept in an environment of temperature < 0℃?
When a colloidal silica binder is subject to freezing, usually a few degrees below the freezing point of water, the water (in fact a solution of water and sodium salts) in the colloidal silica will start to freeze. The water molecules freeze from the coldest area (usually starting from the surface of the liquid) inwards.
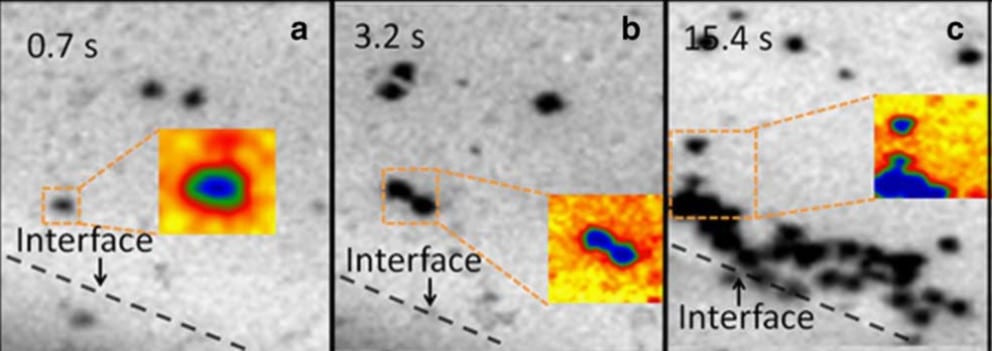
As the water molecules start to crystallise they push the colloid particles away from the solidification front (see Figure 1). This increases the colloid concentration in the unfrozen area and increases the possibility of colloidal sol aggregation the precursor of gelation.
Once agglomeration occurs it is irreversible even when the temperature of the binder is increased back to room temperature (~20 °C). However, if this issue is noticed early enough (before aggregation occurs) then the silica particle can be re-dispersed and aggregation can be avoided. Any aggregation occurred during the freezing process will reduce the concentration of freely dispersed colloidal silica and thus reduces the free binding surfaces.
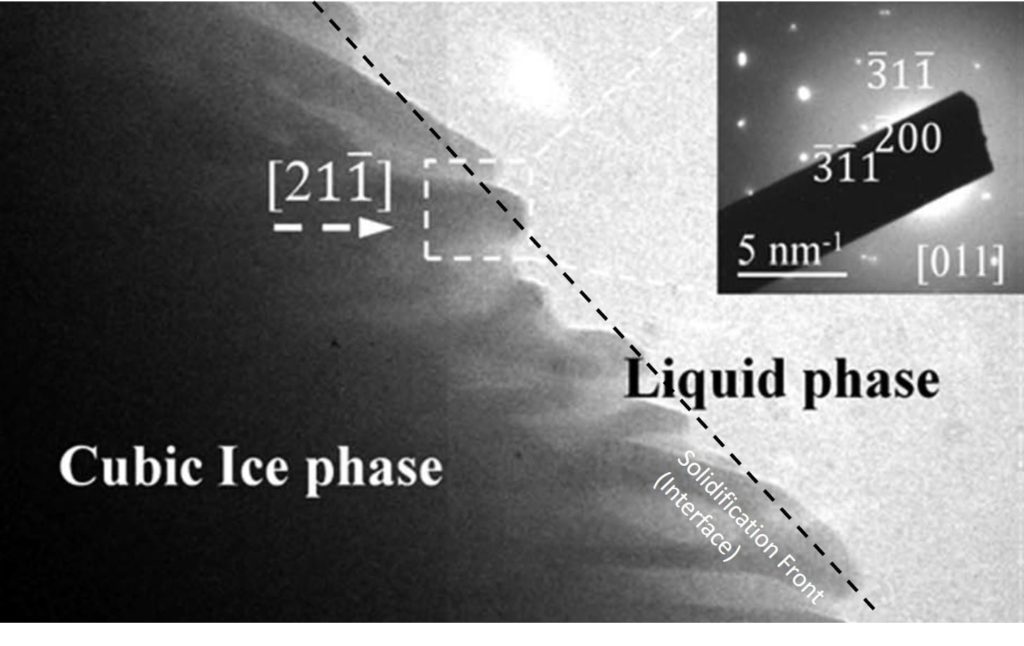 Figure 1: Advancing of Ice crystals during solidification (1).
Figure 1: Advancing of Ice crystals during solidification (1).
Figure 2: Colloid particles being push out of the advancing solidification interface and starts to agglomerate (1).
At the same time as water freezes, it releases latent heat and changes the equilibrium of the system, as a result it is not easy to know the condition of a binder after 5,12, 24 hours under freezing conditions. This depends on other factors including what is the temperature of the binder prior to exposing it to subzero, wind chill effect, total mass of binder exposed to extreme condition etc.
We recommend that colloidal silica should always be stored in temperatures above 2 °C (35 °F). Freezing temperatures cause irreversible aggregation of the silica. Where possible drums and bulk storage tanks should be stored in a heated location, where this is not possible, they should be stored in a location that will protect them from freezing conditions. Tanks that must be located outside should be provided with external heating and lagging. The temperature of the colloidal silica must be kept high enough so that the air in the tank stays above freezing; otherwise, icicles and frost may form above the liquid. While it is necessary to prevent freezing, it’s not desirable to store colloidal silica for lengthy periods at high temperatures. Continuous exposure to high temperatures will shorten the shelf life, avoid storing the product in areas where the temperature routinely goes above 43 °C (110 °F).
Common signs that the binder has been exposed to subzero conditions
After the silica sol has aggregated and precipitated, the process is not reversible. In occasion when the binder exposed to extreme conditions, the binder will not flow smoothly and has agglomerated particles in it even after returning to above 0 °C. The appearance is like sand or ice sludge in water (see photo below).
Process impacts of a binder that has been exposed to subzero conditions
A binder that has begun to aggregate, will show very typical tests on QC / Goods Inward Testing. The pH will be lower, and if left longer to aggregate the silica content can be lower too. In gross cases there may even be a precipitate at the bottom of the drum or IBC where the silica has precipitated out of suspension. A binder that exhibits a low pH and reduced silica content should not be used.
Testing a colloidal silica binder that has been subjected to subzero temperatures
All binder packaging will have a freeze indicator attached to it once it has been produced. The indicator will show if the packaging has been exposed to temperature below 0 °C (see the image below). If the indicator shows that the binder has been in subzero conditions consult your REMET representative prior to use. It is also a good practice to place a temperature data logger in the location where the colloidal silica is kept.
However, if the product is in an IBC or drum, even though the freeze indicators are activated, the product may not have been affected by subzero due to the large binder volume. To be certain, the binder viscosity, silica content and pH test can be used to assess the bulk effect on the material.
We also suggest that to make sure there is no aggregation of colloidal silica occur during the freezing condition, a sample can be taken from the IBC and centrifuged. The supernatant from the centrifuge can be used to determine the percentage of free suspended colloidal silica available. A low silica percentage is an indication that the colloidal sol has been through a sufficiently long freezing condition and the sol is not suitable for use.
The impact of temperatures between 1 °C and 5 °C on a colloidal silica binder
Binder performance is not impacted by temperature under 5 °C providing the material has not frozen (Remember that the freeze indicator only indicates that the material has been exposed to freezing conditions, not that the material is frozen or damaged). The impurities/ ions in colloidal silica reduce the freezing point of a pure water. However, there are a few simple quality control tests that can be carried out to assure if the colloidal silica is good to be used, for example Brookfield binder viscosity test, Potentiometric titrator (surface area of colloidal silica) or simple pH meter and silica content test.
For more information on testing your colloidal silica material please contact your REMET representative.
References
1. In situ cryogenic transmission electron microscopy for characterising the evolution of solidifying water ice in colloidal systems. Tai, K., Liu, Y. and Dillon, S.J. 20, s.l. : Microscopy and Microanalysis, 2014, Vols. pp. 330-337.
Disclaimer
Information and/or recommendations based on information received are believed to be reliable. Offered free of charge for use by persons with technical skills, at their own discretion and risk, without guarantee of accuracy REMET makes no warranties, express or implied, and assumes no liability as to the use of its products or of any information pertaining thereto.